




See the science
for yourself
Delivering a smoke-free future










contentS

5
6
04 Smoke-free alternatives
for adult smokers
06 Integrating risk reduction and acceptance for current smokers
1
3
2
22 Reduced emissions of harmful chemicals
26 No adverse effect on the
overall indoor air quality
30 Effects on the risk of COPD and CVD
31 Effects on the risk of lung cancer
34 Reduced exposure to harmful chemicals
36 Positive impact on smokers’ health
39 Use behavior
40 Majority of THS users no longer smoke cigarettes and use THS exclusively
41 Smokers who have switched to PMI heated tobacco products (HTPs) worldwide
42 Real-world data on hospitalizations before and after launch of heated tobacco products
4
platform
development
See page 20
toxicological
assessment
See page 28
clinical
assessment
See page 33
perception
and behavior
See page 38
Long-term
Assessment
See page 40
PMI SCIENCE Delivering a smoke-free future

01




In this section
04 Smoke-free alternatives
for adult smokers
06 Integrating risk reduction and acceptance for current smokers


1

Our goal at Philip Morris International (PMI) is to offer smoke-free alternatives that have the potential to reduce the risk of developing smoking-related diseases as compared with continued smoking. Recent advances in science and technology have made it possible to develop innovative products that current adult smokers accept and that are less harmful alternatives to continued smoking.
PMI SCIENCE Delivering a smoke-free future
02

03


04

05

PMI SCIENCE Delivering a smoke-free future








Point of intervention
switching
to smoke-free
product



Time

Smoking tobacco causes a number of serious diseases and increases the risk of early death.
Tobacco control strategies in most countries focus on supply and demand measures intended to prevent initiation, reduce consumption and encourage cessation. These measures have resulted in a decline in smoking prevalence over the last three decades, but are unlikely to quickly eliminate smoking altogether. In fact, based on population trends, it is estimated that there will be more than 1 billion smokers by 2025. About the same number as today.
Given the number of smokers who will continue to smoke cigarettes, and as the technology now exists and will continue to develop, it makes sense to offer them less harmful, yet satisfying, smoke-free alternatives.
In this regard, sensible risk-based policies and regulations should allow adult smokers to access scientifically substantiated smoke-free products to help address the harm caused by smoking more effectively and rapidly than traditional policy measures alone.
SMOKE-FREE ALTERNATIVES
FOR ADULT SMOKERS
Chapter 1

Smoking and cessation

We know from epidemiology that smoking increases the risk of developing a smoking-related disease.
Epidemiology has also demonstrated that if a smoker quits, the risk
of developing a smoking-related disease decreases.
Since smoking cessation is the “Gold Standard” for assessing the reduction in risk for adult smokers,1 our goal is to develop products that have a risk profile as close as possible to that of smoking cessation, while being acceptable alternatives to cigarettes for adult smokers who would otherwise continue to smoke.
Figure 1
Conceptual depiction of the cumulated risk of smoking and the effect of cessation over time.2 Note that the straight lines used in this figure are for illustration purposes only as the accumulation of disease risk and the reduction upon cessation and switching to a smoke-free product follow different trajectories for specific diseases.

“Seven years ago, we set
out to create a new future for PMI—a future in which cigarettes would be obsolete, replaced by less harmful, science-based alternatives.”
Jacek Olczak Chief Executive Officer, Philip Morris International
06

07

PMI SCIENCE Delivering a smoke-free future


Combustion
No Combustion
1 Combustible products include cigarettes, cigars, and cigarillos
2 Non-combustible products include ENDS, HTPs, pouches, snus
3 NRT products include patches, gums
ENDS: Electronic Nicotine Delivery Systems, HTPs: Heated Tobacco Products, NRT: Nicotine Replacement Therapy



Cigarettes & other combustible products 1


Non-
combustible products2


Nicotine
replacement
therapy
products3




Cessation


For any smoke-free
alternative to be successful
in reducing population harm,
it has to fulfil two criteria:
it must be scientifically proven to be significantly less harmful than cigarettes; and, it should be satisfying for current
adult smokers.
In addition to taste, and other sensory aspects, a nicotine profile approaching that of cigarettes is important in achieving acceptance by adult smokers. Experts, including the U.S. Surgeon General and the U.K. Royal College of Physicians, agree that nicotine, while addictive, is not the primary cause of smoking-related diseases.
Smoking-related diseases, such as lung cancer, cardiovascular disease, and emphysema, are caused primarily by inhaling harmful compounds largely formed when tobacco is burned, not by nicotine alone.
As the U.S. Food and Drug Administration (FDA) has stated3 “inhalation of nicotine (i.e., nicotine without the products of combustion) is of less risk to the user than the inhalation of nicotine delivered by smoke from combusted tobacco products.”
Not all tobacco products are the same. Their use exists along a continuum of risk, where smoking combusted tobacco yields the highest risk and quitting nicotine and tobacco products altogether contributes the lowest risk. The use of other tobacco products, including smoke-free products, also lie in that continuum of risk. Products that don’t burn tobacco are likely to be far less harmful alternatives to continued smoking.
INTEGRATING RISK REDUCTION AND
ACCEPTANCE FOR CURRENT SMOKERS
Chapter 1

Smoke-free product regulation

Advanced technological and scientific solutions require advanced regulatory tools.
Offering smoke-free alternatives to adult smokers can reduce the risk of harm. This approach should be supported by public health bodies, in addition to existing efforts to prevent smoking initiation and encourage cessation.
Progressive regulatory oversight can protect public health whilst simultaneously ensuring that adult smokers can access smoke-free products as well as accurate and
non-misleading information
about them.
Access to such information is a common-sense approach to
public health.
Modern regulation should take into account the novel nature of smoke-free products. It should ensure that specific quality and performance standards are met. Robust scientific evidence should demonstrate their reduced-risk profile.
Our contribution so far
Our comprehensive body of scientific evidence for our leading smoke-free product, the tobacco heating system (THS), has been submitted to regulatory bodies in several countries.
We submitted Modified Risk Tobacco Applications (MRTPAs) in December 2016 and Premarket Tobacco Product Applications (PMTAs) in March 2017
to the U.S. FDA.
We also submitted technical and scientific dossiers to regulatory authorities in several EU member states.
In April 2019, following a rigorous science-based review through the PMTA pathway, the U.S. FDA determined that authorizing
THS for the U.S. market is appropriate for the protection of the public health.4
Figure 3
The continuum of risk shows that non-combustible products are lower risk than cigarettes.
Figure 2 The Harm Reduction equation
Adapted from Clive Bates’ presentation at the E-Cigarette
Summit on 19 November 2013.







realized
public
health
benefit
Reduced
Risk
Product
acceptance
and Usage


In July 2020, the U.S. FDA authorized
the marketing of the Tobacco
heating system as a modified risk tobacco product with reduced exposure information.5
The agency found that the issuance of the modified risk tobacco product orders with reduced exposure information would be “appropriate
to promote the public health and
is expected to benefit the health
of the population as a whole.”



Our smoke-free products are in various stages of development, production or commercialization; all designed to offer better alternatives for adult smokers than continuing to smoke. All newly developed products undergo rigorous testing, including nonclinical and clinical assessment. This booklet summarizes the key scientific results on the tobacco heating system (THS).
2
PMI SCIENCE Delivering a smoke-free future
08

09



11

10


PMI SCIENCE Delivering a smoke-free future
Inhalable
nicotine products
Non-inhalable
nicotine products
Chapter 2


The products depicted are subject to ongoing development and therefore visuals are illustrative and do not necessarily represent the latest stages of product development.
All of these products avoid burning tobacco or producing smoke, each in its own way. They are designed to deliver
a nicotine-containing aerosol with a reduced level
of harmful and potentially harmful toxicants compared
to cigarette smoke.
Instead of producing an
aerosol that is inhaled, these products are placed under
the lip so that nicotine can
be absorbed via the mouth.
Our non-inhalable products include oral tobacco products and oral nicotine products.

Tobacco heating system (THS)
THS heating technologies include blade and induction heating.
THS uses an electronic heat-control technology to heat tobacco within a specific temperature range. Extensive laboratory and clinical data are available supporting its potential for risk reduction compared with continued cigarette smoking. Standard THS consists of three components: a pocket charger, a holder, and a heated tobacco unit.

Electrically heated tobacco products
MESH vaping system (MVS)
MVS are battery-powered devices that vaporize a nicotine-containing liquid, scientifically engineered to give a consistent user experience, without
the limitations of a coil and
wick system.
E-vapor products
Aerosol heating system (AHS)
AHS combines elements of e-cigarettes and heated tobacco products into a single hybrid system. Developed by KT&G.
Aerosol heated
tobacco products


Nicotine pouches
The oral smokeless category doesn’t involve a device, heating, or the inhalation of an aerosol. Instead,
a teabag-like pre-portioned pouch
that contains nicotine (but not tobacco), is placed between the gum and lip and removed after use. The nicotine is extracted through the action of saliva and is absorbed mainly via the mucous membranes
in the mouth before entering the bloodstream. Some nicotine can also reach the gastrointestinal tract if saliva is swallowed.
Oral smokeless
pouches

“No one product will address all adult smokers’ or nicotine users’ individual preferences. OUR APPROACH IS TO DEVELOP A RANGE OF SMOKE-FREE PRODUCTS, SO THAT SMOKERS WHO WOULD OTHERWISE CONTINUE TO SMOKE CIGARETTES CAN FIND A SUITABLE ALTERNATIVE THAT ALLOWS THEM TO FULLY SWITCH.”

Pin-based heating system (PHS)
PHS electrically heats the tobacco
using either inductive or resistive
heating. Developed by KT&G.*

* KT&G is the leading tobacco and nicotine company in South Korea.




Our scientific assessment is built on a collaborative approach and expertise in the fields of chemistry, toxicology, biology, informatics, medicine, and perception and behavior. Our practices are inspired by the pharmaceutical industry and aligned with U.S. FDA’s
Draft Guidance for Modified Risk Tobacco Product Applications (2012).6
3
PMI SCIENCE Delivering a smoke-free future
12

13
































our scientific approach
Chapter 3


The assessment of a smoke-free product’s risk reduction potential relies on the quality
of the initial product design and on strict manufacturing controls to ensure that the product delivers a consistent aerosol.
The products are specifically designed with the aim to eliminate or reduce the levels of Harmful and Potentially Harmful Constituents (HPHCs) found in their aerosol compared to those found in cigarette smoke.
In this initial phase of designing a product, it is verified that the product’s design does not pose any additional risks to those already known for combustible cigarettes. Only then can we begin to conduct further research.
Toxicological assessment
aims to confirm whether
the reduced formation of HPHCs leads to reduced toxicity and reduced risk of smoking-related diseases in laboratory models.
PMI conducts a series of in vitro and in vivo studies on smoke-free products, following Good Laboratory Practice (GLP), to determine whether the reduced levels of HPHCs lead to a reduced toxicity compared with cigarette smoke.
We take toxicological assessment one step further by using a new area of science known as systems toxicology. Systems toxicology helps determine whether reduced toxicity leads to reduced risk of smoking-related diseases in laboratory models.
Clinical studies are a cornerstone
of our assessment program.
They help determine the extent to which adult smokers would find the product an acceptable alternative to cigarettes.
They assess whether a reduction in
the formation of HPHCs measured
in the laboratory leads to a reduction in HPHC exposure under real-world conditions when an adult smoker switches to the product.
And they also investigate whether switching from cigarettes to a
smoke-free product has a beneficial effect on a smoker’s health profile
by reducing the risk of smoking-
related diseases as compared to continued smoking.
platform
development
toxicological
assessment
clinical
assessment
Pharmacokinetics /
Pharmacodynamics
Measures
 Smoking behavior
Smoking behavior
 Nicotine uptake
Nicotine uptake
 Subjective effects
Subjective effects
1
Reduced exposure
Measures
 Exposure to
Exposure to
harmful smoke constituents
2
Exposure response
Measures
 Changes in blood
Changes in blood
chemistry
 Functional health
Functional health
and symptoms
3



Perception and behavior studies help us evaluate risk perceptions and patterns of use of smoke-free products among various adult consumer groups.
For smoke-free products to have an overall positive impact on public health, it is important that adult smokers use them, that those intending to quit tobacco and nicotine altogether are not dissuaded by these products, and that nonsmokers do not use them.
Moreover, smokers should understand that quitting is the best way to reduce smoking-related health risks, and
that these products are only for smokers who would otherwise continue to smoke.
The assessment of our smoke-free products continues after the products are placed
on the market.
Long-term assessment, including post-market studies, will confirm whether these products reduce the risk of smoking-related diseases such as chronic obstructive pulmonary disease, cardiovascular disease
and lung cancer.
We combine a number of approaches, including safety surveillance, clinical studies and epidemiological studies,
in order to obtain a progressively clearer picture of the risk-reduction potential of our products.
PERCEPTION
and BEHAVIOR
LONG-TERM
ASSESSMENT


2
3
Safety reports
 Feedback from consumers
Feedback from consumers
 Through scientific literature
Through scientific literature
cohort studies
 Defined group
Defined group
of people
 Followed through time
Followed through time
Cross-sectional
surveys
 Defined groups
Defined groups
of people
 Snapshots in time
Snapshots in time
1
14

15

PMI SCIENCE Delivering a smoke-free future


QUALITY PRINCIPLES
At each step, scientific rigor is applied to generate data that may support a claim that smoke-free products reduce exposure to harmful and potentially harmful constituents and present less risk of harm than continued smoking.
A risk-based Quality Management System has been conceived for smoke-free products to coordinate and guide activities with the aim of ensuring quality and integrity of the product during its complete lifecycle, from the conception through to commercialization.
HOW DOES PMI CONDUCT
ITS SCIENTIFIC ASSESSMENTS?





























Product Design
and Control
Quality by Design (QbD)7
Nonclinical Studies
OECD Test Guidelines; ICH Guidelines; FDA Guidelines; Applicable National Regulations; GLP; ISO 17025
Clinical Studies
WMA Declaration of Helsinki12;
Based on ICH-GCP E6 (R2)13; Applicable Local Regulations
Perception and Behavior Assessment
Based on GEP-DGEpi14; Based on FDA Guidance on TPPI; FDA Guidance on PRO15; Applicable National Regulations; Declaration of Helsinki
Observational Studies
IEA GEP16; Applicable
National Regulations
Aerosol Chemistry
OECD GLP8; ISO9 17025; ICH
Q2 (R1)10; ISO 3308*, 3402,
4387*, 8454, 10315:2013,
10362-1*, 13110, 19290,
20768, 20778





platform
development
toxicological
assessment
clinical
assessment
PERCEPTION
and BEHAVIOR
LONG-TERM
ASSESSMENT
Indoor Air Quality
ISO 17025; EN 1525111;
ISO 15593, 18144, 18145,
16814, 16000-6, 11454
Chapter 3

ensuring quality and integrity of the product during its complete lifecycle, from the conception through to commercialization.

* With slight modifications needed to adapt to smoke-free products.
16

17

PMI SCIENCE Delivering a smoke-free future








highlights


4
In the following section we dive into many of our key findings.
From the vital discovery that there is no burning in THS (version 2.2) through to the findings around positive impact on smokers’ health up to the usage pattern of THS when the product is launched.

































platform
development
toxicological
assessment
clinical
assessment
PERCEPTION
and BEHAVIOR
LONG-TERM
ASSESSMENT
40 Majority of THS users no longer smoke cigarettes and use THS exclusively
41 Smokers who have switched to PMI heated tobacco products (HTPs) worldwide
42 Real-world data on hospitalizations before and after launch of heated tobacco products
PMI SCIENCE Delivering a smoke-free future
18

19


There is no burning IN THS
Chapter 4: platform development
We have conducted several studies to demonstrate the absence of combustion in
THS, including temperature measurements, experiments demonstrating the absence
of net exothermic processes, and measurements of constituents that represent typical markers of combustion.18
Our studies also support that the aerosol of THS does not contain solid particles that are produced when tobacco is burned.19 In addition, since burning requires oxygen, we have tested THS in an oxygen-free atmosphere.
The results showed that oxygen does not play a major role in the thermochemical degradation of the THS tobacco or the aerosol formation. Combustion does not occur during THS use.
Decades of scientific research show that the primary cause of smoking-related disease is the high levels of Harmful and Potentially Harmful Constituents (HPHCs) in
smoke formed during the combustion of tobacco.
During a puff of a cigarette, the temperature increases to more than 800 °C at the tip.17 The combustion of tobacco results in the formation of smoke (containing high levels of HPHCs), heat, and ash.


Figure 4
Our heated tobacco technologies include blade heating and induction heating.
The induction THS uses the same internal tobacco heating principle as the blade THS, but without the blade. This means there is no direct contact between the electronics and the heating element.
The tobacco is heated from within the tobacco stick through energy transfer to a heating element via a magnetic field.

Induction

A heating blade that heats the tobacco plug in the consumable radially outwards from the center of the tobacco plug.

Blade

Heated tobacco




Did you know?
Smoke is a result of combustion

For combustion to occur,
three things need to be present: a fuel to be burned,
a source of oxygen and a
source of heat.
Heated tobacco products do not combust tobacco and therefore produce an aerosol that is not smoke.
combustion
Heat

O2
Oxygen
Fuel

Smoke contains high levels of harmful chemicals,
most of which are formed during combustion, also known as burning.

Smoke
Heat, Light
and ASH

During the puff of a cigarette, the temperature increases to more than 800°C at the tip. The combustion of tobacco results in the formation of ash, heat and light (energy), and smoke.
Cigarettes







COMBUSTION DOES NOT OCCUR DURING THE USE OF THE TOBABCCO HEATING SYSTEM.
20

21

PMI SCIENCE Delivering a smoke-free future

* Average reductions in levels of a range of harmful chemicals (excluding nicotine) compared to the smoke of a reference cigarette (3R4F). Based on the FDA 18,21 IARC,22 and WHO 923 lists of HPHCs.
Reduced emissions of
brharmful chemicals

Aerosol chemistry:
Targeted analysis
We measured a number of harmful chemicals in the aerosol of THS and compared them to the levels found in the smoke of a standard reference cigarette (3R4F). On average, a 95% reduction in the levels of these HPHCs in THS aerosol was observed.20
Figure 5
Reduced emissions of HPHCs from
THS use. The average level of HPHCs in
THS aerosol is shown by the red bar and is compared with the average level of HPHCs in smoke from the 3R4F reference cigarette marked as 100% in the graphic.
Chapter 4: platform development continued
Aerosol chemistry:
Untargeted analytical screening
The comprehensive chemical characterization of THS aerosol using untargeted analytical screening methods revealed that a total of 532 chemical constituents (including water, glycerin, and nicotine, which were measured using different methods) were present at concentrations ≥ 100 ng/heated tobacco unit.24
The identities for 80% of all chemical constituents measured using untargeted screening, representing > 96% of the total determined mass, were confirmed with purchased reference chemicals. All compounds that were detected in THS aerosol ≥ 100 ng/heated tobacco unit were also found to be present in smoke from the standard reference cigarette 3R4F.
Only a minority of compounds in THS aerosol were present at concentrations exceeding those measured in cigarette smoke.
To identify any potential new hazards presented by exposure to THS
aerosol, untargeted differential screening was also performed, which only looked for chemicals that were significantly more concentrated in THS aerosol compared with cigarette smoke.
The compounds that were found to be significantly higher in THS aerosol compared with cigarette smoke, including three compounds that were unique to THS aerosol (all with concentrations ≥100 ng/heated tobacco unit), were submitted for toxicological evaluation. Four compounds were subsequently highlighted to be of potential toxicological concern.
The levels of these four compounds were very low and the U.S. FDA
concluded that “Although some of the chemicals are genotoxic or cytotoxic, these chemicals are present in very low levels and potential effects are outweighed by the substantial decrease in the number and levels of HPHCs found in combusted cigarettes.”25
Figure 6
This graph presents the untargeted characterization results of the regular variant
of the THS heated tobacco unit. The 532 chemicals present in THS aerosol are also present in 3R4F cigarette smoke (≥ 100 ng/heated tobacco unit).















































































532 chemicals in THS aerosol
ca. 4800
chemicals in 3R4F smoke



reference
cigarette
THS

95%*
Average reduction

100%

0
5%



BY ELIMINATING COMBUSTION, THE LEVELS OF HARMFUL CHEMICALS ARE REDUCED ON AVERAGE BY 95% IN THE AEROSOL OF THE TOBACCO HEATING SYSTEM COMPARED TO THOSE IN CIGARETTE SMOKE.


5000

0

4500

4000

3500

3000

500

1000

1500

2000

2500

Cigarette
THS

To the best of our knowledge, this is the first time that such a comprehensive in-depth chemical characterization of the aerosol composition of a heated tobacco product has been reported. This work represents several years of effort in the field of analytical method development and advanced structural identification techniques, which have been applied to the THS aerosol.
22

23

PMI SCIENCE Delivering a smoke-free future

Chapter 4: platform development continued


Figure 7
The picture shows the visual difference between the particulate matter of standard reference cigarette
smoke (1R6F, left) and the particulate matter of THS aerosol (right) after collection on Cambridge glass-fiber pads (1 stick per product, aerosol regime ISO 20778:2018).
Did you know?















cigarette
THS
Cambridge
GLASS-FIBER pads
People have been talking about tar in cigarettes for a long time, yet ‘What is tar?’ is a question often asked today, and there are many myths surrounding the topic.
Tar is the weight of solid and liquid residue in cigarette smoke, after nicotine and water have been removed. It’s not an added chemical nor the material used to pave roads, it’s simply a weight measurement. So, is tar measurement useful? If we only take the weight into account, no it’s not.
Out of context, the weight gives no indication of residue content nor the risk of harm because the level of toxicants within that weight are unknown, and the World Health Organization (WHO) agrees, “While several Parties include tar in their regulatory policies, it is not on the priority list of toxicants in tobacco smoke emissions, as the composition of tar varies qualitatively and quantitatively in each type of product, limiting the possibility for validated testing and measurement.”26
Put another way, the WHO says,
“Tar need not be measured, as it is not a sound basis for regulation,
and the levels can be misleading.”
When people consider the risk of harm of a product in relation to tar it’s more important to look at the content of the residue rather than its weight. When we look at the content of cigarette smoke, there are thousands of chemicals released and of those the FDA has listed 93 known toxicants. It is the presence of and exposure to these chemicals that plays a role in the development of smoking-related diseases, not the tar measurement.


TAR NEED NOT BE MEASURED, AS IT IS NOT A SOUND BASIS FOR REGULATION, AND THE LEVELS CAN BE MISLEADING.
World Health Organization
24

25

PMI SCIENCE Delivering a smoke-free future


* The classification of toxicants was based on the list of the International Agency for Research on Cancer and the FDAʼs established list of Harmful and Potentially Harmful Constituents (HPHCs), 2012. In addition, other compounds were measured too.

Figure 8
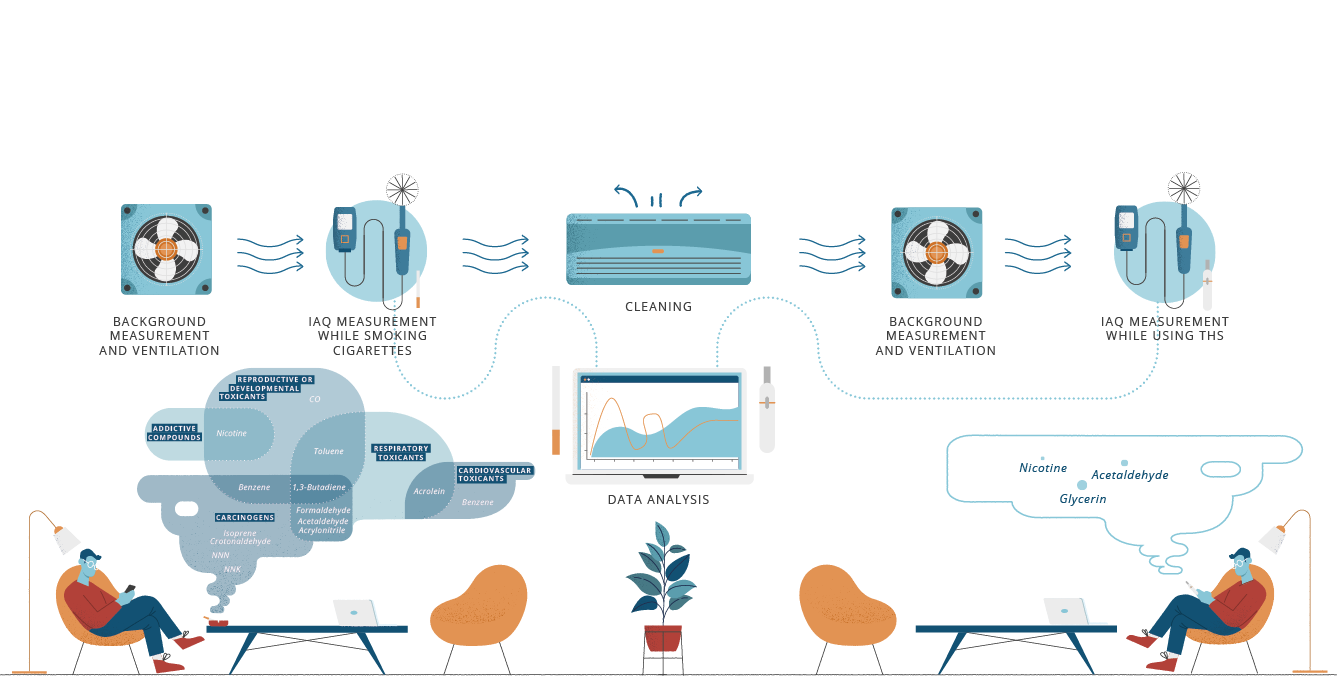
When THS was used indoors, out of 24 measured compounds, only nicotine, acetaldehyde and glycerin were measured at levels higher than the background, although well below the exposure limits established in air quality guidelines. The use of THS in an indoor environment, where regulatory norms of adequate ventilation are respected, does not adversely affect the overall indoor air quality.
cigarette
Tobacco heating system (THS)
*

Measuring air quality markers in accordance with international guidelines allows to assess the quality of indoor air.
We measured 24 compounds including carbonyls, tobacco-specific nitrosamines, and volatile organic compounds under simulated residential conditions.
No adverse effect on the
overall indoor air quality

THS has no adverse effect on the overall indoor air quality (IAQ)
Chapter 4: platform development continued


THE USE OF THE TOBACCO HEATING SYSTEM IN AN indoor environment, where regulatory norms of adequate ventilation are respected, does not adversely affect the overall indoor air quality.
When using THS, the levels of 21 of these compounds did not increase beyond the levels already present as background in our dedicated Indoor Air Quality room. Only the nicotine, acetaldehyde and glycerin were measurably higher than the background, although well below
the exposure limits established in
air quality guidelines.27, 28, 29
26

27

PMI SCIENCE Delivering a smoke-free future



STUDIES SHOW A SUBSTANTIAL REDUCTION IN TOXICITY OF THE AEROSOL OF THE TOBACCO HEATING SYSTEM COMPARED TO CIGARETTE SMOKE.
We have conducted a series
of regulatory toxicology tests
to compare the toxicity of
THS aerosol with that of the smoke from a standard
reference cigarette (3R4F).
In our laboratories, we observed
a substantial reduction in toxicity
of THS aerosol compared to
cigarette smoke.20

Figure 9
The chart shows our findings concerning the relative in vitro toxicity of THS aerosol compared with the smoke from the 3R4F reference cigarette using three in vitro assays (Neutral Red Uptake, Ames and Mouse Lymphoma) commonly used to assess cytotoxicity and genotoxicity.
Reduced toxicity
Chapter 4: toxicological assessment

reference
cigarette



100%

0

THS
10%


5%


 95%
95%
Average reduction


Not
mutagenic

>90%
Average
reduction


Cytotoxicity
Bacterial
Mutagenicity
mammalian
genotoxicity
28

29

PMI SCIENCE Delivering a smoke-free future

Figure 11
Regarding the disease endpoint lung cancer, the charts are showing the findings on combined lung adenoma and/or adenocarcinoma incidence (A) and multiplicity (B) from a carcinogenicity study in A/J mice exposed to either 3R4F cigarette smoke or THS aerosol for up to 18 months. Gene analysis confirms similarities between spontaneously developed tumors and tumors developed in THS aerosol-exposed animals.
A study was conducted to compare carcinogenic effects of THS aerosol with 3R4F cigarette smoke over the lifetime (18 months) of A/J mice.
The A/J mouse spontaneously develops tumors in the lungs, but they occur more often and the number is higher in mice that are exposed to cigarette smoke. In this study, the number of mice who developed tumors (incidence) and the number of tumors per animal (multiplicity) were significantly lower in THS aerosol exposed mice than in those exposed to cigarette smoke. Incidence and multiplicity were similar in the mice exposed to fresh air and those exposed to THS aerosol.31, 32
Chapter 4: toxicological assessment continued
Figure 10
The charts are showing the findings for the disease endpoints COPD and CVD in a mouse switching study. Lung emphysema (A) and atherosclerotic plaque volume (B) were measured in Apoe-/- mice that were exposed for 8 months to either 3R4F smoke or THS aerosol. A group of mice was first exposed for two months to 3R4F smoke and then switching to either THS aerosol or fresh air. The fresh air control is also depicted here. Lung emphysema scores were assessed by histopathology after 8 months of exposure, atherosclerotic plaque volumes were measured by micro-CT after 7 months of exposure.
effects on the risk of lung cancer
effects on the risk of COPd and CVD



PMI conducted a systems toxicology study in an animal model (Apoe-/- mouse) that develops atherosclerotic plaques and emphysema when exposed to cigarette smoke.
In this study, mice were exposed to either the smoke of a standard reference cigarette (3R4F) or THS aerosol for 8 months. A group of mice was first exposed for two months to 3R4F smoke and then randomized to either THS aerosol (switching) or fresh air (cessation). Switching to THS aerosol following two months of cigarette smoke led to reduced impact on biological mechanisms and disease endpoints associated with COPD and CVD in a manner similar to smoking cessation.30

EXPOSURE TIMELINE











Cigarette
START
month 15
month 18
Air
THS (low concentration)
THS (medium concentration)
THS (high concentration)
THS (high concentration)
Air
Reduced risk of lung cancer
Lung tumor incidence (%)
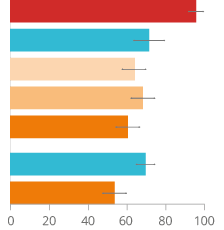
(A) incidence
Lung tumor Multiplicity

(B) Multiplicity

SWITCHING TO THE TOBACCO HEATING SYSTEM LED TO reduced impact
on biological mechanisms and disease endpoints associated with COPD and CVD compared to continued smoking in laboratory models.

UNLIKE CIGARETTE
SMOKE, THE AEROSOL
OF THE TOBACCO
HEATING SYSTEM DOES NOT LEAD to increased lung tumor incidence and multiplicity in
a mouse model.
EXPOSURE TIMELINE












Cigarette
START
month 2
month 8
Cigarette
Cigarette
THS
Air
Cessation
THS
Reduced risk of COPD and CVD
Lung Emphysema
score
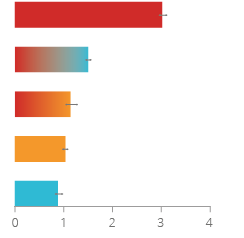
(A) COPD
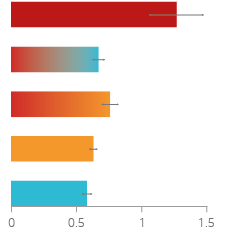
atherosclerotic
plaque volume (mm3)
(B) CVD

30

31

PMI SCIENCE Delivering a smoke-free future


Figure 12
After three weeks, THS aerosol caused
no obvious discoloration to the teeth
and no color mismatch between
the teeth and dental resins, unlike cigarette smoke.
Nicotine occurs naturally in the tobacco plant and at significantly lower levels in some other plant varieties. When tobacco is burned, nicotine is transferred to the smoke.
Nicotine can be acutely toxic when ingested or absorbed at levels much higher than what consumers are exposed to when using tobacco or nicotine-containing products, and it
can increase a person’s heart rate
and blood pressure.
When tobacco smoke is inhaled, nicotine is absorbed through the lungs into the bloodstream, and reaches the brain in about 10-20 seconds. There, nicotine binds to specific receptor molecules, mimicking the actions of a naturally occurring brain chemical, acetylcholine. In turn, these activated receptors influence the brain’s ‘pleasure center,’ which may explain the subjective pleasurable effects associated with smoking, but also relates to the potential for dependence.
Nicotine also affects other parts of the body such as the heart and blood vessels. The physiological effects of nicotine on the brain and body are
short term and reversible.
“It is primarily the toxins and carcinogens in tobacco smoke
– not the nicotine – that cause
illness and death.”
U.K. National Institute for Health and Care Excellence (NICE), Smoking: harm reduction; Public health guideline (2013)
Nicotine used in pharmaceutical products (nicotine replacement therapies (NRTs)) as well as in e-cigarettes is usually extracted from tobacco. It is possible to produce synthetic nicotine, but the process
is costly. Certain people should not
use products that contain nicotine.
Minors should not use or have access
to tobacco or nicotine-containing products. Nicotine products should
not be used by non-nicotine users.
Nicotine-containing products should also not be used during pregnancy or while breast-feeding. Nicotine-containing products should not be used by people who have or are at risk of heart disease, are diabetic, are epileptic or experience seizures. Experts, including the U.S. Surgeon General and the U.K. Royal College of Physicians, agree that, while nicotine is addictive and not risk-free, it is not the primary cause of smoking-related diseases.
Smoking-related diseases, such as lung cancer, cardiovascular disease, and emphysema, are caused primarily by inhaling harmful chemicals largely formed when tobacco is burned, not
by nicotine alone.
Did you know?

We have shown in the pharmacokinetic and pharmacodynamic clinical studies that the level of
nicotine, and the timing of its peak concentration in the
blood, were comparable for smokers and for subjects
who switched to THS.
Furthermore, the urge-to-smoke scores were similar for smokers and switchers. This suggests that switchers do not seek to use THS more frequently than smokers seek to use cigarettes, and that switchers can find THS acceptable and satisfying.34
Chapter 4: toxicological assessment continued
Less staining
on teeth

Chapter 4: clinical assessment
Nicotine uptake


THE AEROSOL OF THE TOBACCO HEATING SYSTEM DISCOLORS TEETH SIGNIFICANTLY LESS THAN CIGARETTE SMOKE.
enamel

composite
resin

Dentin

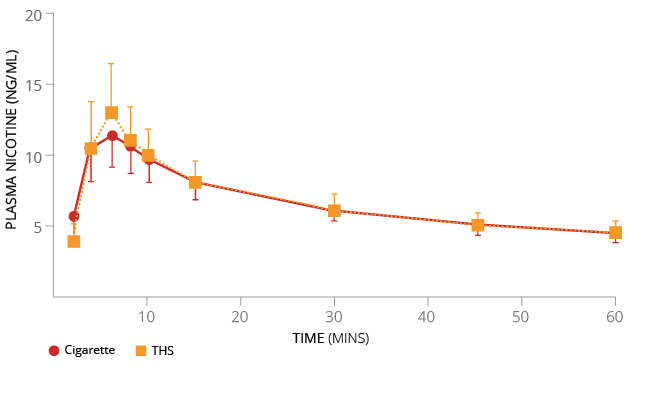
Figure 13
Nicotine plasma concentration over 60 minutes for a cigarette and THS.




THS
Cigarette smoke
Our studies on human teeth demonstrate the reduced discoloration effects of THS aerosol compared to the smoke of a 3R4F cigarette.
Teeth that had cavities filled with dental resins were exposed to cigarette smoke or THS aerosol for four days a week, followed by brushing and incubation. After three weeks of such exposure, cigarette smoke exposure caused an overall larger color change compared with THS aerosol exposure. Exposure to cigarette smoke also caused mismatches between the
tooth and the dental resins while
THS aerosol exposure did not.33
Please, find our verification
programs here:
www.intervals.science
www.intervals.science/resources/sbv-improver
Colors depicted in this figure
are only representative.
32

33

PMI SCIENCE Delivering a smoke-free future

In our 5-day and 90-day clinical reduced exposure studies,
we measured biomarkers in the blood and urine representing exposure to selected harmful chemicals.
We found that levels of 15 biomarkers of exposure in participants switching completely to THS were comparable to the levels of those who quit smoking for the duration of the study. In both cases, the levels remained significantly below those observed in subjects who continued smoking during the study.35, 36
Chapter 4: clinical assessment continued
Figure 14
The effects of switching to THS or smoking abstinence on biomarkers of exposure levels for 4 selected HPHCs. We examined a total of 15 biomarkers, all displaying the same trend. The same type of study in the U.S. (NCT01989156) showed comparable results.
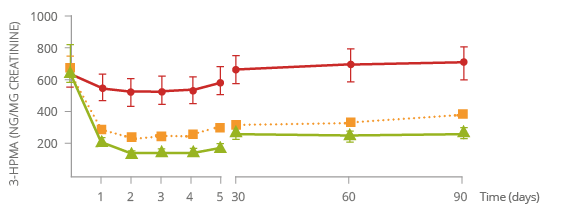
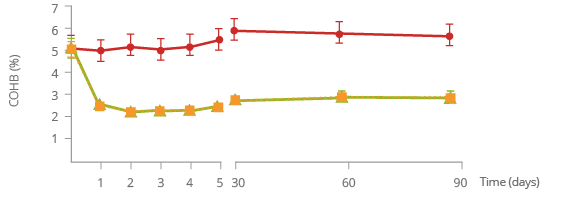
Reduced exposure to harmful chemicals







1,3-butadiene*

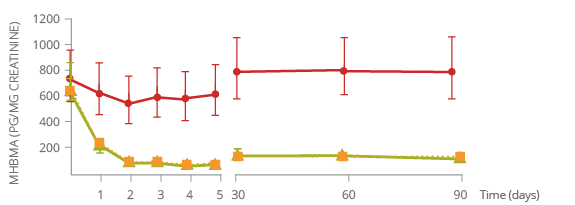
Acrolein*
Carbon Monoxide*

SMOKERS SWITCHING COMPLETELY TO THE TOBACCO HEATING SYSTEM WERE EXPOSED to significantly lower levels of harmful chemicals compared
to those who continued smoking during the study.
* Levels of biomarkers detected
(3-month study in Japan).
BENZENE*
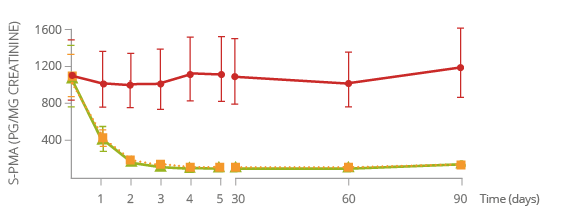


S-Phenylmercapturic
acid (S-PMA) is a biomarker of benzene exposure.


Carboxyhemoglobin (COHb) is a biomarker of carbon monoxide exposure.

34

PMI SCIENCE Delivering a smoke-free future

Did you know?
Figure 15
In the U.S. FDA established list of harmful and potentially harmful constituents (HPHCs)

Monohydroxybutenyl mercapturic acid (MHBMA) is a biomarker of 1,3-butadiene exposure.

1,3-Butadiene
is classified as a carcinogen, respiratory toxicant, and reproductive or developmental toxicant.
Acrolein
is classified as
respiratory and cardiovascular toxicant.
Carbon monoxide
is classified as a
reproductive or developmental toxicant.
Benzene
is classified as a carcinogen, cardiovascular toxicant, and reproductive or developmental toxicant.

3-Hydroxypropylmercapturic
acid (3-HPMA) is a biomarker of acrolein exposure.

35

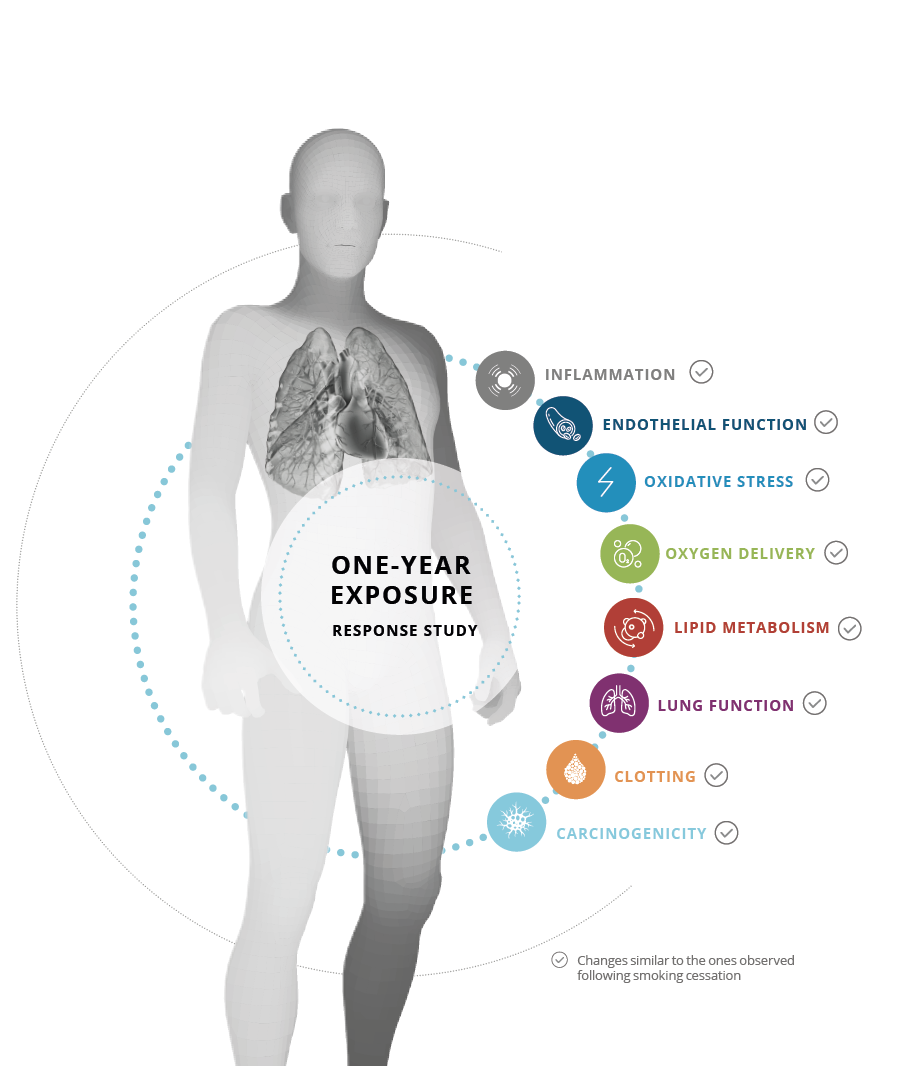
Positive impact on smokers’ health

We conducted a one-year exposure response study, which consisted of a six-month study followed by a six-month extension study. Eight biomarkers of potential harm (BoPH) covering different physiological pathways altered by cigarette smoking were measured with the aim to demonstrate favorable changes on all eight BoPH (as observed when stopping smoking) when switching from smoking to THS versus continued cigarette smoking. To compare the effect of switching to THS to that of smoking cessation, we conducted in parallel a one-year smoking cessation study evaluating the same core set of eight BoPH.
Our studies showed that smokers who switched from cigarettes to THS for 12 months had favorable changes in all eight BoPH, in the same direction as upon smoking cessation. We found that in predominant THS users, the lesser the concomitant cigarette smoking the greater were the favorable effects on the BoPH. Furthermore, exclusion of the highest intensity of cigarette consumption in THS users showed that the favorable response to THS use corresponded to more than 67% of that of smoking cessation for seven of the eight BoPH.37, 38, 39


Chapter 4: clinical assessment continued

Figure 16
Changes in BoPH related to smoking-related diseases when switching to THS.

CLINICAL FINDINGS PROVIDE EVIDENCE ON THE POTENTIAL OF THE TOBACCO HEATING SYSTEM TO REDUCE THE RISK OF DEVELOPING CVD, COPD, AND CANCER COMPARED WITH CONTINUED CIGARETTE SMOKING.




































































![THS-Cigarette Difference [absolute values (95% CI)]](assets/images/item_14518.png)








Month
5
4
3
2
1
0
-1
3
6
12
3
6
12
3
6
12

HDL-C
(mg/dL)
FEV1
(%pred)
WBC
(GI/L)

![Relative Reductions (THS vs. Cigarette) [% (95% CI)]](assets/images/item_14400.png)














Month
3
6
12
3
6
12
3
6
12
3
6
12
60
50
40
30
20
10
0
-10
3
6
12
sICAM-1
(mg/dL)
11-DTX-B2
(pg/mgcreat)
8-epi-PGF2α
(pg/mgcreat)
Total NNAL
(pg/mgcreat)
COHb
(%)














36

37

PMI SCIENCE Delivering a smoke-free future


Chapter 4: perception and behavior
INTENTION to use
USE BEHAVIOR

We conducted perception and behavior studies to investigate the effect of introducing
smoke-free products on:
1. Tobacco use behavior amongst adult smokers
2. Tobacco use initiation amongst adult nonsmokers (i.e., former smokers and never smokers)
3. Consumer understanding of product messages and perception of risks
Our pre-market perception and behavior studies conducted in the U.S. showed that substantial proportions of current adult smokers (up to 39%) expressed intention to use THS. At the same time, our perception and behavior studies showed that low proportions of nonsmokers expressed intention to use THS (i.e., adult former smokers ≤6.4%, adult never smokers and legal age to 25-years-old never smokers ≤1.1%). Moreover, we also conducted studies for THS with adults who were not consumers of tobacco or nicotine-containing products (TNPs) in several countries in 2020 and 2021. The results of those studies also show very low intention to use THS among adult former users of TNPs and adults who had never used TNPs. The study findings also showed that adult smokers correctly understand that switching to THS presents less risk of harm than continued cigarette smoking, while not being risk-free. Furthermore, our data showed a low impact of THS communication materials on the intention to quit all tobacco among adult smokers with the intention to quit smoking.
Our pre-market, actual use perception and behavior study conducted in the U.S. aimed to measure the effect of THS on tobacco use behavior among adult daily cigarette smokers. This six-week observational actual use study showed that 15% of smokers switched from cigarettes to THS exclusive or predominant use.40 The study findings also showed that the availability of THS did not lead to an increase in total tobacco product consumption (THS and cigarettes).
OUR ACTUAL USE PERCEPTION AND BEHAVIOR STUDIES SHOWED THAT A SIZEABLE PROPORTION OF SMOKERS WERE LIKELY TO SWITCH FROM CIGARETTES TO THE EXCLUSIVE OR PREDOMINANT USE OF THE TOBACCO HEATING SYSTEM.


The U.S. actual use study findings were aligned with similar pre-market studies conducted in Germany, Italy, South Korea, Japan, and Switzerland which showed that between 10% and 38% of smokers switched from cigarettes to THS exclusive or predominant use. We have conducted post-market studies after the commercialization of THS (under the brand IQOS) and the study results show that these pre-market actual use studies well predicted THS future use behavior.
PMI will continue to conduct perception and behavior studies as part of PMI’s overall scientific assessment program for PMI’s smoke-free products.
OUR PERCEPTION AND BEHAVIOR STUDIES SHOWED THAT SMOKERS CORRECTLY UNDERSTAND THAT SWITCHING TO THE TOBACCO HEATING SYSTEM PRESENTS LESS RISK OF HARM THAN CONTINUED CIGARETTE SMOKING.

38

39

PMI SCIENCE Delivering a smoke-free future

Moreover, these studies show that more than half of THS users no longer smoke cigarettes and use THS exclusively. This shift towards the use of smoke-free products suggests that smoke-free products, such as THS, are acceptable alternatives to cigarette smoking.
Our post-market cross-sectional surveys also show very low to non-existing TNP initiation with THS among never TNP users (<0.1%). More than 99% of current THS users have a history of TNP use before switching to THS, and only 1% to 2% of current THS users relapsed or re-initiated tobacco use with THS.
On a population level, based on the results of our post-market cross-sectional surveys, the commercialization of THS appears to be in line with the principles of tobacco harm reduction.
PMI will continue to conduct post-market studies as part of PMI’s overall scientific assessment program for PMI’s smoke-free products.
As part of our ongoing long-term assessment, we have conducted repeated post-market cross-sectional surveys in a representative sample of the adult population from Japan41, 42, Italy, and Germany, to monitor the use prevalence of THS after its commercialization.
These studies show that the total use prevalence of tobacco or nicotine-containing products (TNPs) in Japan, Italy, and Germany is overall stable across time, with higher total TNP use prevalence in Italy and Germany compared to Japan. These studies also show a growing prevalence of THS use among TNP users across time with a higher prevalence of THS use in Japan (2019: 18.4%) compared to Italy (2019: 4.1%) and Germany (2019: 1.2%).
MAJORITY OF THS USERS NO LONGER SMOKE CIGARETTES AND USE THS EXCLUSIVELY




Figure 18
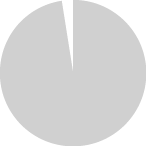
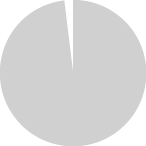
Tobacco relapse or re-initiation among current THS users
with a history of TNP use in Japan, Italy, and Germany in 2019.
Smokers
Ex-smokers ≤12 months (relapse)
Ex-smokers >12 months (re-initiation)
Cigarettes or other TNP
THS
Figure 17
History of TNP use in THS users in Japan, Italy, and Germany in 2019.

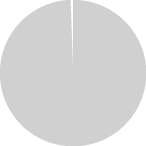

99.5%
0.5%
Italy



99.3%
0.7%
Japan



99.8%
0.2%
Germany


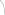


97.5%
1.5%
Japan
1.0%

Italy
0.9%



98.2%
0.9%
1.5%

Germany
0.9%



97.6%
Chapter 4: Long-term Assessment

* Users of PMI HTPs, for which PMI heated tobacco units (HTUs) represented at least a portion of their daily tobacco consumption over the past 7 days.
** For markets where there are no HTPs other than PMI HTPs, daily individual consumption of PMI HTUs represents the totality of the users’ daily tobacco consumption in the past seven days. For markets where PMI HTPs are among other HTPs, daily individual consumption of HTUs represents the totality of the users’ daily tobacco consumption in the past seven days, of which at least 70% is PMI HTUs.
*** Source: PMI Financials or estimates, Tobacco heating system (THS) user panels and PMI Market Research. Status as of June 2022. Note: Russia and Ukraine not included in total users.

More than 99% of
current THS users have a history of TNP use before switching to THS.
Only 1% to 2% of current THS users relapsed or
re-initiated tobacco use with THS.
40

41

PMI SCIENCE Delivering a smoke-free future

Figure 19
Number of smokers who have switched to PMI HTPs worldwide***.
As of March 2022, there are approximately 17.9 million* users of PMI HTPs globally, excluding Russia and Ukraine, of which approximately 12.7 million** (71%) have switched to PMI HTPs and stopped smoking.
Smokers who have switched to PMI heated tobacco products (HTPs) worldwide

Total users
Estimated users who have switched to PMI HTPs and stopped smoking









Q1
5
5
15
20
Q1
Q2
Q2
2021
2022
Q3
Q4








As heated tobacco products (HTPs) become available in different markets, it is important to assess their impact on the health of the individual and the population as a whole. In the absence of long-term epidemiological studies, real-world data can be used to look for early signals of the population health impact of introducing HTPs.
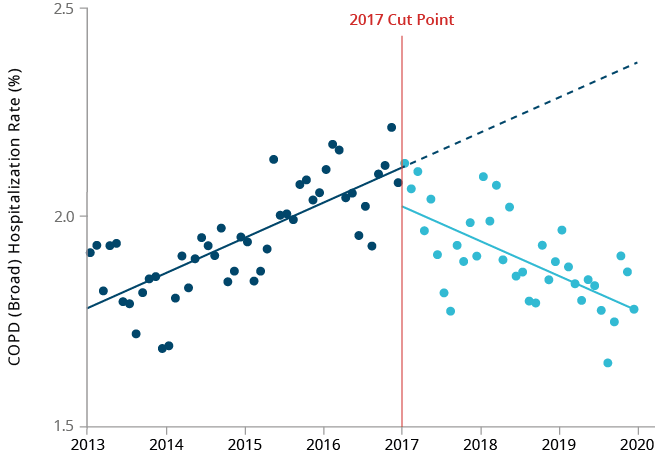
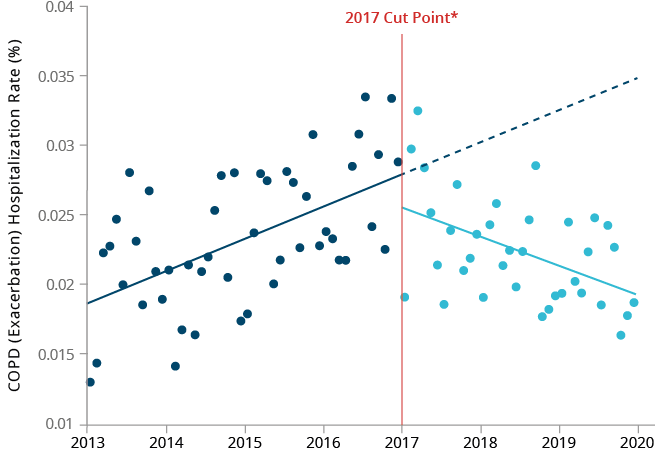
We explored the temporal association between the introduction of HTPs in Japan and hospitalization rates for selected smoking-related diseases using two different Japanese databases. We performed time trend analyses to compare the observed versus expected rates of hospitalizations for COPD, COPD exacerbations and ischemic heart disease (IHD) before and after the introduction of HTPs (including our THS) in Japan. Although the frequency of COPD exacerbations is low in Japan, analyses using the Medical Data Vision (MDV) database showed a significant reduction in the rate of hospitalization due to COPD exacerbations after the introduction of HTPs in Japan.
We also observed a small decline in the hospitalization rate for IHD, although not significant. Analyses using the Japan Medical Data Center (JMDC) database showed a significant reduction in the number of hospitalizations for COPD (all codes) when using all available data, and for IHD when using diagnosis procedure combination (DPC) only data.43 Non-significant declines in hospitalizations for COPD exacerbation + lower respiratory tract infections and IHD were also observed using all JMDC data. It should be noted, however, that the absolute count of hospitalizations due to COPD exacerbation, as well as for all COPD endpoints with DPC-only data in the JMDC database, were extremely small; hence, no meaningful interpretation could be drawn.
The minor differences between the results of these two studies may be attributable to the differences in the demographic characteristics of the two database populations. Nevertheless, both studies showed some positive changes in smoking-related disease hospitalization trends following the introduction of HTPs in Japan. There are many limitations to this type of study,
and it is important to acknowledge that the results do not indicate a causal relationship.
Real-world data on hospitalizations before and after launch of heated
tobacco products
Chapter 4: Long-term Assessment continued
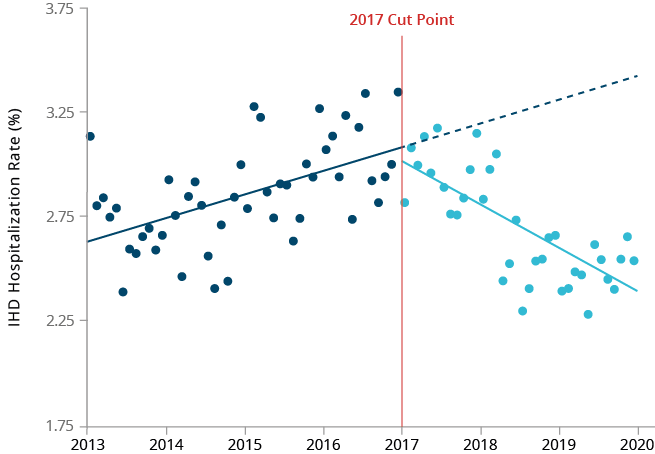
Figure 22
IHD hospitalization rate over time in Japan, using DPC data from JMDC database.
Figure 21
COPD (all codes) hospitalization rate over time in Japan, using all available data from JMDC database.
Pre-HTP
Post-HTP
Prediction from Pre-HTP data

Pre-HTP
Post-HTP
Prediction from Pre-HTP data

* The 2017 cut point was based on sales data, since HTPs were introduced into
the Japanese market end of 2014, it could be assumed there is a two-year transition period in order to have a relevant market exposure to HTPs.

Hospitalization rates for COPD and IHD started decreasing shortly after
the launch of heated tobacco products in Japan.
Figure 20
COPD exacerbation hospitalization rate over time in Japan, using MDV database.
Pre-HTP
Post-HTP
Prediction from Pre-HTP data





42

43

PMI SCIENCE Delivering a smoke-free future




5

Independent studies and reviews
Dutch National Institute
for Public Health and the Environment (RIVM)

All-Russia Scientific Research Institute for Tobacco and Tobacco products

National Institute of Public Health Japan

Superior
Health Council
of Belgium

U.k. Committee
on Toxicity (COT)

Public Health
England (PHE)
U.S. Food and Drug Administration
(U.S. FDA)

German Federal
Institute for Risk
Assessment (BfR)

National Institute of Cardiology Ukraine

China National Tobacco Quality Supervision and Test Center

In this section
Over the last few years, numerous independent studies have already confirmed different elements of
our research on the tobacco heating system (THS).
PMI SCIENCE Delivering a smoke-free future
44

45



Independent studies
and Reviews
Chapter 5
Public Health
England (PHE)44
2018: Public Health England (PHE) published a review of the evidence on e-cigarettes and heated tobacco products, and stated that heated tobacco products likely reduce users’ and bystanders’ exposure to harmful compounds compared to cigarettes.
PHE also stated available evidence suggests that heated tobacco products may be considerably less harmful than tobacco cigarettes and more harmful than e-cigarettes.
U.k. Committee
on Toxicity (COT)45
2017: The U.K. Committee on Toxicity conducted a review of available evidence on two heated tobacco products, one of which
is IQOS*, and concluded that these products “are likely to reduce risks for smokers.”
Dutch National Institute
for Public Health and the Environment (RIVM)
2018: RIVM published a fact sheet on novel tobacco products that are heated and an English-language summary.
They concluded that “The use of Heatsticks with the IQOS is harmful to health, but probably less harmful than smoking tobacco cigarettes,” based on their aerosol chemistry measurements, which are “of the same order of magnitude as in the data of Philip Morris.” 46
2020: RIVM published the findings of its research on “A Method for Comparing the Impact on Carcinogenicity of Tobacco Products: A Case Study on Heated Tobacco Versus Cigarettes.”
RIVM developed a method to estimate risk – or assess the potential magnitude of the health impact – between tobacco products.
In their publication they assessed eight carcinogens to understand the likely health impact on individuals who switch to IQOS, compared to those who continue smoking.
In their conclusions they state that – while IQOS is not risk-free – it is associated with 10 to 25 times lower exposure to these carcinogens, and that this could translate
into a substantially improved risk profile.47
German Federal
Institute for
Risk Assessment (BfR)48
2018: The German Federal Institute for Risk Assessment (BfR), published laboratory studies on IQOS in Archives of Toxicology, finding that reductions in selected toxicants measured by the institute “are likely to reduce toxicant exposure.”
U.S. Food and Drug Administration
(U.S. FDA)
2018: The U.S. FDA , in a briefing document, reviewed PMI’s data supporting IQOS and the available independent literature about IQOS. The briefing document included a section explaining the results of the FDA’s IQOS aerosol chemistry measurements.49
2019: Following a comprehensive assessment
of PMI’s premarket tobacco product applications, the U.S. FDA confirmed that IQOS is appropriate for the protection of public health and has authorized it for sale in the United States. “Appropriate for the protection of public health” means that looking at population as a whole, new products cannot pose the same or greater harm to public health as smoking. The FDA published a detailed report describing their assessment and their conclusions including results on aerosol chemistry, toxicology and unintended use.4
2020: The U.S. FDA issued decisions on PMI’s Modified Risk Tobacco Product (MRTP) applications for the IQOS tobacco heating system. In doing so, the agency found that
the issuance of the modified risk tobacco product orders with reduced exposure claims would be “appropriate to promote
the public health and is expected to benefit
the health of the population as a whole.”
This decision follows a review of the
extensive scientific evidence package
PMI submitted to the FDA in December
2016 to support its MRTP applications.5

To date, over 50 studies from independent laboratories have results that are in line with our findings on THS.
Researchers working for the American Cancer Society confirm that the introduction of IQOS
is the only likely cause of cigarette sales decline in Japan.50
Research by the Japanese Department of Environmental Health, National Institute of Public Health, compared selected chemicals
in the aerosol generated by IQOS and in smoke from reference cigarettes. The research shows significant reductions in the levels of several chemicals, in line with those found by PMI’s research.51
The China National Tobacco Quality Supervision and Test Centre, a member of the WHO Tobacco Laboratory Network, published an independent study comparing the harmful chemicals present in IQOS aerosol and cigarette smoke, which generally agree with PMI’s results.52
One of Ukraine’s leading research institutes conducted a 6-month clinical study on IQOS, which was published in prominent national medical periodical Ukrainian Health, showing no significant adverse effect on users of smoke-free products.53
Researchers at the University of St. Andrews, Scotland, calculated that IQOS aerosol has “lower cancer potencies than tobacco smoke by at least one order of magnitude, but higher potencies than e-cigarettes.”54
The first independent study investigating levels of carbon monoxide in the exhaled breath (eCO) after use of two recently marketed heated tobacco products (HTPs) was conducted by Pasquale Caponnetto, Marilena Maglia, Gaetano Prosperini, Barbara Busà and Riccardo Polosa and was published in Respiratory Research. The aim of this randomized cross-over study was to measure the exposure levels of the combustion marker, eCO of subjects after use of two HTPs and to compare these levels with participants’ own brand of cigarettes. The study found no eCO elevations during inhalational testing with HTPs under investigation in any of the study participants.55
Research by cardiologist and leading e-cigarette researcher Dr. K. Farsalinos on IQOS was published in the journal Addiction, showing that IQOS emits lower levels of carbonyls than a commercial cigarette,
but higher levels than an e-cigarette.56

Many government bodies
have conducted literature reviews or performed research on scientifically substantiated heated tobacco products, finding that they expose users to significantly lower levels
of harmful chemicals.
* IQOS is the brand name under which the tobacco heating system is commercialized.
46

47

PMI SCIENCE Delivering a smoke-free future




In this section
Our value creation model describes what we do and how we allocate our resources to deliver long-term value for both our company and our stakeholders.
Our mission
Accelerate the end of smoking.
WHAT WE DO
Replace cigarettes with less harmful tobacco and nicotine products for the benefit of adults who would otherwise continue to smoke.
6
+980
scientists, engineers, technicians and support staff working on our
smoke-free products.
30
scientific and engineering disciplines including: materials science, consumer electronics, clinical science and systems toxicology.
1,770
patents granted for smoke-free technologies by the five largest intellectual property offices in the world (IP5, cumulative)*.
USD 120 million
invested in construction
of the Cube, PMI’s remarkable R&D facility in Neuchâtel (Switzerland).
USD 617 million
total R&D expenditure.
99%
of total R&D expenditure was dedicated to smoke-free products in 2021.
USD 9.2 billion
invested by PMI since 2008 in the development, scientific substantiation, manufacturing, commercialization and continuous innovation for smoke-free products.
30
worldwide locations
with research and
technology partners.
452
scientific publications by PMI since 2008 – most open access.
*IP5 jurisdicitions are Europe (patents granted by the European Patent Office), China, South Korea, Japan and the U.S.
PMI SCIENCE Delivering a smoke-free future
48

49



see the facts for yourself
Chapter 6
* MRTP: Modified Risk Tobacco Product.
** PMI conducted a consumer test of its CHTP in the last quarter of 2021. As a result of the feedback, the design of the company’s current technology has been discontinued. PMI is assessing alternative designs for this consumer segment.
*** Excluding Russia and Ukraine.
Opening of a new R&D center in Switzerland, focusing exclusively on smoke-free products.

Opening of a new research laboratory in Singapore.
Spinning-off from Altria Group Inc. PMI enhances R&D to develop smoke-free alternatives to cigarettes.
Acquisition of the technology for Nicotine Salts Product (NSP).

Release of the U.S. FDA draft guidance on the submission of an MRTP* application, PMI’s assessment approach largely in line with it.

Agreement between PMI and Altria to commercialize the Tobacco Heating System (THS) and Carbon Heated Tobacco Product (CHTP)** in the U.S. subject to issuance of FDA marketing orders.

Launch of THS commercialized as IQOS in Nagoya (JP) and Milan (IT).

Launch of PMIScience.com to publicly share with scientific community methodologies and findings related to PMI‘s smoke-free products.









PMI announces ambition to “convince all current adult smokers that intend to continue smoking to switch to smoke-free products as soon as possible”.
Submission of an MRTP application for THS to the U.S. FDA, which upon issuance of marketing orders would allow relative risk claims in comparison with cigarettes.
Launch of a new corporate website reflecting PMI’s commitment to transition away from cigarettes to smoke-free products.
Pilot testing of CHTP in the Dominican Republic.

Launch of MESH Vaping System (MVS) in London (U.K.).

U.S. FDA authorizes the sale of THS 2.2 (IQOS 2.4 version) in the U.S.



Our aim is that 40 million cigarette smokers will have switched to our smoke-free products.


Launch of a new THS 3 using Smartcore Induction System, commercialized
as IQOS ILUMA.
U.S. FDA authorizes marketing of THS 2.2 (IQOS 2.4 version) as a Modified Risk Tobacco Product concluding that switching completely to THS reduces body’s exposure to harmful and potentially harmful chemicals.







U.S. FDA issues an MRTP order authorizing PMI to market also the THS 2.2 (IQOS 3 version) with reduced exposure information.
Approximately 17.9 million*** users of PMI HTPs globally, of which approximately 12.7 million (71%) have switched to PMI HTPs and stopped smoking.

50

51

PMI SCIENCE Delivering a smoke-free future

2009
2010
2011
2012
2008
2014
2015
2013
2017
2018
2019
2020
2021
2016

2022


Glossary and acronyms
Aerosol
An aerosol is a suspension of fine solid particles and/or liquid droplets in a gas (usually air). Cigarettes generate a smoke aerosol that is the result of the combustion (burning) of tobacco and contains carbon-based solid particles. While smoke is an aerosol, not all aerosols are smoke.
PMI’s smoke-free products do not produce smoke because they do not burn tobacco. Instead, they generate a nicotine-containing aerosol, either by heating tobacco or through other technologies that do not involve combustion.
Consumers typically use the term “vapor” to refer to the aerosol generated from heated tobacco products or other nicotine-containing products.
BIOMARKER OF POTENTIAL HARM
Biomarkers of potential harm
(BoPH) are a measurable change
in biochemical, physiological
(organs, tissues, cells), or behavioral function within an organism that is known to be associated with a health impairment or disease.
These biomarkers indicate the body’s response to exposure to harmful chemicals. While BoPH
do not necessarily cause these health concerns, their presence
and magnitude help identify whether
a person already has
or is in danger of developing a
health impairment or disease.
Biomarkers of exposure
Indicate exposure to a potentially hazardous substance. In our case, the biomarker may be a cigarette smoke constituent or metabolite that is measured in a biological fluid or tissue. Biomarkers of exposure can provide a measure of internal dose, which is the amount of the constituent taken up into the body.
COMBUSTION
Combustion is the process of burning a substance in oxygen, producing heat and often light. When a cigarette is lit, the combination of tobacco (fuel) and oxygen in the air generates a self-sustaining combustion process that consumes the tobacco. The combustion of tobacco results in the formation of smoke (that contains a range of chemical constituents), heat, and ash. The high heat associated with combustion leads to the thermal breakdown of the tobacco when it is burned, resulting in the production of many of the toxicants found in cigarette smoke.
STANDARD REFERENCE CIGARETTE (e.g., 2R4F,
3R4F, 1R6F)
Standard reference cigarettes such as the 2R4F, 3R4F, and 1R6F reference cigarettes, are provided by the University of Kentucky for laboratory testing. A standard reference cigarette is used as a consistent and uniform test item for nonclinical investigations by tobacco manufacturers, contract and government laboratories, and academic institutions. The current version is the standard reference cigarette 1R6F.
U.S. FDA
United States Food and Drug Administration
COPD
Chronic Obstructive Pulmonary Disease.
CVD
Cardiovascular disease.
Exposure
Exposure to a chemical describes how much of a chemical comes into contact with a human, laboratory animal or cell culture so that it might be inhaled, ingested or absorbed and how often and for how long
that does happen. Exposure of humans to chemicals is important
to understand because it may have an influence on human health.
HPHCs
Harmful and Potentially Harmful Constituents, HPHCs are chemicals or chemical compounds in tobacco products or tobacco smoke that cause or could cause harm to smokers or nonsmokers. The Food, Drug and Cosmetic Act (FD&C Act) requires tobacco manufacturers and importers to report the levels of HPHCs found in their tobacco products and tobacco smoke.
MODIFIED RISK TOBACCO PRODUCT (MRTP)
‘Modified risk tobacco product’ or ‘MRTP’ is the term formally used by the U.S. Food and Drug Administration to describe an alternative to cigarettes that is associated with less risk of disease.
Mutagenic
In genetics, a mutagen is a physical or chemical agent that changes the genetic material, usually DNA, of an organism and thus increases the frequency of mutations above the natural background level.
Pharmacodynamics
Pharmacodynamics is the study of the response of the body to a pharmacological compound.
Pharmacokinetics
Pharmacokinetics is the study
of the process by which a pharmacological compound is absorbed, distributed, metabolized and eliminated by the body.
References
52

53

PMI SCIENCE Delivering a smoke-free future

Institute of Medicine (2012). Scientific standards for studies on modified risk tobacco products. The National Academies Press, Washington, DC.
Smith, M. R., et al. (2016). Evaluation of the Tobacco Heating System 2.2. Part 1: Description of the system and the scientific assessment program. Regul Toxicol Pharmacol 81 Suppl2: S17-S26.
U.S. Department of Health and Human Services. Food and Drug Administration. (2016). Deeming Tobacco Products To Be Subject to the Federal Food, Drug, and Cosmetic Act, as Amended by the Family Smoking Prevention and Tobacco Control Act; Restrictions on the Sale and Distribution of Tobacco Products and Required Warning Statements for Tobacco Products. Available at www.federalregister.gov/d/2016-10685/p-155.
U.S. Food and Drug Administration (2019). Premarket Tobacco Product Marketing Order TPL (Technical Project Lead Review); PM0000424-79.
U.S. Food and Drug Administration (2020).
Scientific Review of Modified Risk Tobacco Product Application (MRTPA) Under Section 911(d) of the FD&C Act -Technical Project Lead.
U.S. Department of Health and Human Services. Food and Drug Administration. Center for Tobacco Products (CTP). (2012). Modified risk tobacco product applications: Draft guidance. Available at www.fda.gov/media/83300/download.
Juran, J. M. (1992). Juran on Quality by Design: The New Steps for Planning Quality into Goods and Services.
OECD Series on Principles of Good Laboratory Practice (GLP) and Compliance Monitoring.
International Organization for Standardization.
ICH – Validation of Analytical Procedures: Text and Methodology.
European Committee for Standardization (2007). CEN European Standard EN 15251. Indoor Environmental Input Parameters for Design and Assessment of Energy Performance of Buildings Addressing Indoor Air Quality, Thermal Environment, Lighting and Acoustics. European Committee for Standardization, Brussels.
World Medical Association (WMA) Declaration of Helsinki – Ethical Principles for Medical Research Involving Human Subjects.
ICH – Guideline for Good Clinical Practice.
Good Epidemiologic Practice (GEP) – GermanSociety for Epidemiology (DGEpi).
Food and Drug Administration (2009). Guidance for Industry – Patient-Reported Outcome Measures: Use in Medical Product Development to Support Labeling Claims.
IEA Guidelines for proper conduct in epidemiologic research (2007).
Baker, R. R. (1975). Temperature variation within a cigarette combustion coal during the smoking cycle. High Temp Sci 7: 236-247.
Cozzani, V., et al. (2020). An experimental investigation into the operation of an electrically heated tobacco system. Thermochim Acta 684: 178475.
Pratte, P., et al. (2017). Investigation of solid particles in the mainstream aerosol of the Tobacco Heating System THS2.2 and mainstream smoke of a 3R4F reference cigarette. Hum Exp Toxicol 36: 1115-1120.
Schaller, J. P., et al. (2016). Evaluation of the Tobacco Heating System 2.2. Part 2: Chemical composition, genotoxicity, cytotoxicity, and physical properties of the aerosol. Regul Toxicol Pharmacol 81 Suppl2: S27-S47.
U.S. Department of Health and Human Services. Food and Drug Administration. Center for Tobacco Products (CTP). Guidance for Industry. Reporting Harmful and Potentially Harmful Constituents in Tobacco Products and Tobacco Smoke Under Section 904(a)(3) of the Federal Food, Drug, and Cosmetic Act. DRAFT GUIDANCE. March 2012. Available at www.fda.gov/media/83375/download
International Agency for Research on Cancer (IARC) group 1 carcinogens. Available at List of Classifications – IARC Monographs on the Identification of Carcinogenic Hazards to Humans (who.int).
World Health Organization (2008). The scientific basis of tobacco product regulation: Second report of a WHO study group. WHO Technical Report Series 951. Available at The Scientific Basis of Tobacco Product Regulation (who.int).
Bentley, M. C., et al. (2020). Comprehensive chemical characterization of the aerosol generated by a heated tobacco product by untargeted screening. Anal Bioanal Chem 412:2675-2685.
U.S. Food and Drug Administration. Premarket Tobacco Product Marketing Order TPL (Technical Project Lead Review); PM0000424-79. 29 Apr 2019; Section 6 – Summary of Toxicological Findings: p42. www.fda.gov/media/124247/download
World Health Organization (2015). WHO study group on tobacco product regulation: Report on the scientific basis of tobacco product regulation: Fifth report of a WHO study group. WHO Technical Report Series, no. 989.
Mitova, M. I., et al. (2016). Comparison of the impact of the Tobacco Heating System 2.2 and a cigarette on indoor air quality. Regul Toxicol Pharmacol 80: 91-101.
Mitova, M. I., et al. (2019). Air quality assessment of the Tobacco Heating System 2.2 under simulated residential conditions. Air Qual Atmos Health 12(7): 807-823.
Nordlund, M., et al. (2019). Scientific substantiation of the absence of Environmental Tobacco Smoke (ETS) emission during use of the Electrically Heated Tobacco System (EHTS). Version 1.0, available on pmiscience.com here.
Phillips, B., et al. (2016). An 8-month systems toxicology inhalation/cessation study in Apoe-/- mice to investigate cardiovascular and respiratory exposure effects of a candidate Modified Risk Tobacco Product, THS 2.2, compared with conventional cigarettes. Toxicol Sci 149(2): 411-432.
Wong, E. T., et al. (2020). Reduced chronic toxicity and carcinogenicity in A/J mice in response to life-time exposure to aerosol from a heated tobacco product compared with cigarette smoke. Toxicol Sci 178(1): 44-70.
Titz, B., et al. (2020). Respiratory effects of exposure to aerosol from the candidate modified-risk tobacco product THS 2.2 in an 18-month systems toxicology study with A/J mice. Toxicol Sci 178(1): 138-158.
Zanetti, F., et al. (2019). Effects of cigarette smoke and tobacco heating aerosol on color stability of dental enamel, dentin, and composite resin restorations. Quintessence Int 50(2): 156-166.
Brossard, P., et al. (2017). Nicotine pharmacokinetic profiles of the Tobacco Heating System 2.2, cigarettes and nicotine gum in Japanese smokers. Regul Toxicol Pharmacol 89: 193-199.
Lüdicke, F., et al. (2018). Effects of switching to the Tobacco Heating System 2.2 menthol, smoking abstinence, or continued cigarette smoking on biomarkers of exposure: a randomized, controlled, open-label, multicenter study in sequential confinement and ambulatory settings (Part 1). Nicotine Tob Res 20(2): 161-172.
Our clinical studies are registered on www.clinicaltrials.gov. The ID of the presented study is: NCT01970995.
Lüdicke, F., et al. (2019). Effects of switching to a heat-not-burn tobacco product on biologically relevant biomarkers to assess a candidate Modified Risk Tobacco Product: A randomized trial. Cancer Epidemiol Biomarkers Prev 28(11): 1934-1943.
The IDs of the presented clinical studies are: NCT02396381, NCT02649556, NCT02432729.
Haziza, C. (2021). Assessing the effects of switching from cigarettes to the Tobacco Heating System relative to smoking cessation on biomarkers of potential harm – Additional evidence on the potential to reduce the risk of smoking-related diseases. Conference presentation at the Society for Research on Nicotine & Tobacco, available on pmiscience.com here.
Roulet, S., et al. (2021). Potential predictors of adoption of the Tobacco Heating System by U.S. adult smokers: An actual use study. F1000Res 8: 214.


References continued
54

55

PMI SCIENCE Delivering a smoke-free future

Afolalu, E. F., et al. (2022). Prevalence and patterns of tobacco and/or nicotine product use in Japan (2017) after the launch of a heated tobacco product (IQOS®): a cross-sectional study. F1000Res 10: 504.
Fischer, K., et al. (2022). Trends in prevalence and patterns of use of a heated tobacco product (IQOSTM) in Japan: A three-year repeated cross-sectional study [version 1; peer review: awaiting peer review]. F1000Res 11: 720.
van der Plas, A., et al. (2022). Ischemic heart disease and chronic obstructive pulmonary disease hospitalizations in Japan before and after the introduction of a heated tobacco product. Front Public Health 10: 909459.
Public Health England (2018). Evidence review of e-cigarettes and heated tobacco products 2018. A report commissioned by Public Health England.
U.K. Committee on Toxicity (2017). Statement on the toxicological evaluation of novel heat-not-burn tobacco products.
Rijksinstituut voor Volksgezondheid en Milieu (RIVM)/National Institute for Public Health and the Environment (2018). Nieuwsoortige tabaksproducten die worden verhit.
Slob, W., et al. (2020). A method for comparing the impact on carcinogenicity of tobacco products: A case study on heated tobacco versus cigarettes. Risk Analysis 40(7): 1355-1366.
Mallock, N., et al. (2018). Levels of selected analytes in the emissions of “heat not burn” tobacco products that are relevant to assess human health risks. Arch Toxicol 92(6): 2145- 2149.
U.S. Food and Drug Administration (2018). FDA Briefing Document – January 24-25, 2018 Meeting of the Tobacco Products Scientific Advisory Committee (TPSAC) on the Modified Risk Tobacco Product Applications (MRTPAs) MR0000059-MR0000061 Philip Morris Products S.A.
Stoklosa, M., et al. (2020). Effect of IQOS introduction on cigarette sales: evidence of decline and replacement. Tob Control 29: 381- 387.
Bekki, K., et al. (2017). Comparison of chemicals in mainstream smoke in heat-not-burn tobacco and combustion cigarettes. J UOEH 39(3): 201- 207.
Li, X., et al. (2019). Chemical analysis and simulated pyrolysis of Tobacco Heating System 2.2 compared to conventional cigarettes. Nicotine Tob Res 21(1): 111-118.
Kvasha, E.A., et al. (2017). Evaluation of the impact of electronic nicotine delivery systems on the risk of cardiovascular diseases based on endothelial function and its determining factors. health-ua.com.
Stephens, W. E. (2018). Comparing the cancer potencies of emissions from vapourised nicotine products including e-cigarettes with those of tobacco smoke. Tob Control 27(1): 10-17.
Caponnetto, P., et al. (2018). Carbon monoxide levels after inhalation from new generation heated tobacco products. Respir Res 19(1): 164.
Farsalinos, K. E., et al. (2018). Carbonyl emissions from a novel heated tobacco product (IQOS): comparison with an e-cigarette and a tobacco



Edition 6.1, July 2022
PMIScience.com is operated by Philip Morris International for the purpose of publishing and disseminating scientific information about Philip Morris International’s efforts to develop and assess products that have the potential to reduce individual risk and population harm associated with tobacco use.
PMI Science
Neuchâtel, Switzerland
www.pmiscience.com
© PMI Science June 2022